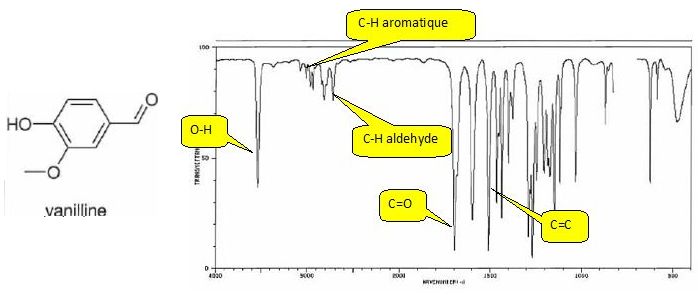
picoSpin spectrometers offer personal convenience without waiting for core facility resources while also providing structure selectivity and high discriminating power in a compact benchtop instrument.Ĭhemical name: (±) -Propranolol hydrochloride (±) -1-Isopropylamino-3- (1-naphthyloxy) -2-propanol hydrochlorideĬoncentration: 11.2% (w / w 0.We develop original concepts which aim at enhancing resolution in spectra of complex samples, to a point where their analysis becomes a faster, easier, and a more accurate process. Peak integration yields quantitative information, further strengthening shift assignments. Shift dispersion and high-resolution means one can discriminate closely spaced proton resonances while also extracting scalar coupling frequencies. These features facilitate spectral assignment of complex molecular structures by producing qualitative spectral quantities. Severe signal broadening suggests slow or intermediate dynamic exchange, as well as expected quadrupolar interaction.Īt 82 MHz the picoSpin 80 1H NMR spectrometer offers the highest field strength in a compact benchtop instrument, delivering chemical shift dispersion and resolution unmatched by other desktop NMR systems. The secondary amine proton (NH) is assigned the broad absorption centered at 9 ppm. 
The hydroxyl proton (OH) assignment to the doublet resonance at 6 ppm is based on peak integration (1H) and multiplicity, having one neighboring proton on C 13. The aliphatic chain protons (C 12-C 13-C 15) are assigned to resonances at 4.13 ppm (2H), 4.39 ppm (1H) and 3.16 ppm (2H), respectively, based on scalar coupling, peak integration, and chemical shift. The isopropyl group gives rise to a doublet methyl signal at 1.25 ppm (C 18, 19 6H), due to coupling to a single methine proton (CH), and an broad multiplet (~3.2 ppm C 17) from the same. Peak integration, however, reveals four protons (4H) contribute to signal intensity. The multiplicity of remaining ring protons, C 3, 4, 7, 8, appearing between 7.25-7.6 ppm, is obscured by signal overlap, making individual assignment difficult. The doublet of doublet multiplicity arises from vicinal 3 J (6 Hz) and long-range 4 J (3 Hz) coupling across the π system. The roughly equal intensity multiplets assigned to protons at C 9, C 6 and C 2 show a 1:1:1 integral ratio and anticipated chemical shifts of 8.25 ppm (1H), 7.8 ppm (1H) and 6.9 ppm (1H), respectively. Anisotropic effects and ring substitution cause the aromatic protons to shift in the region from 6.7 to 8.3 ppm. Hydroxyl (OH) and the secondary amine (NH) also produce distinct signals in the spectrum.Ĭhemical shift assignment of the naphthyloxy proton resonances is aided by peak integration. Despite the complexity of the spectrum, propranolol can be viewed as containing 3 spin systems: a fused-ring aromatic (C 10H 7O), an aliphatic chain (-CH n-) and an isopropyl ( i-Pr) group. 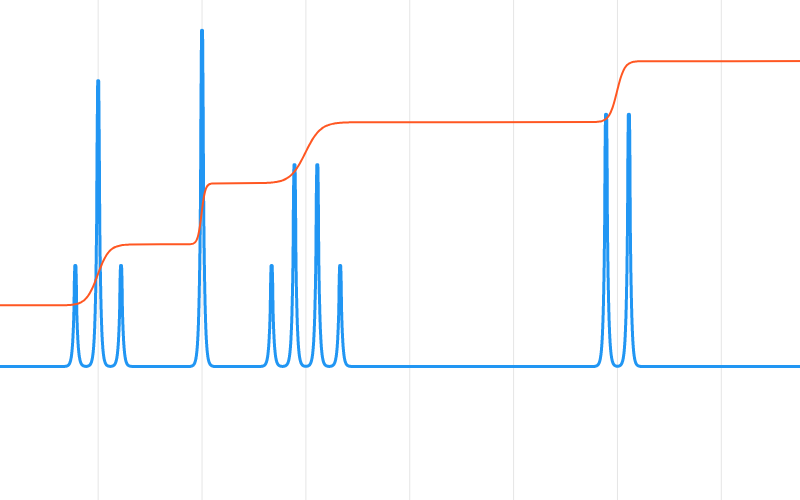
Only the downfield 13C satellite signal (3.33 ppm 0.55% intensity) overlaps analyte signals. The solvent, DMSO, produces a strong resonance at 2.50 ppm, which is used as a chemical shift reference. The 1H NMR spectrum of a 11.2% (w/w 420 mM) solution of propranolol in protonated DMSO was measured at 82 MHz using the Thermo Scientific picoSpin 80 spectrometer. 
It is prescribed medication for treatment of hypertension, severe headaches, and cardiac related dysrhythmia and ischemia. HCl), a nonselective beta-blocker, is representative of a class of drugs that target the beta receptor.(±)-Propranolol hydrochloride (C 16H 21NO 2
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
